Humans are by nature conservative in the sense that we are most comfortable when our environment is stable. Change is frequently viewed as negative. One type of environment that is of conservation concern to European naturalists is the cultural landscape, particularly small wood pastures with scattered or bordering deciduous brush or trees, which are no longer in use for animal husbandry or hay production. These types of habitats have high biodiversity conservation value (Kunttu et al 2019), but provide low economic return, so they have at times been planted to spruce for timber production. In British Columbia, we tend to be more concerned with the preservation of old-growth forests and natural wetlands. In both cases we are trying to preserve the landscape as we know it. We are most comfortable when in a familiar environment.

Nature is not static, however. It is in constant flux, with organismal shifts in a constant cycle that keeps ecosystems healthy. Abiotic and biotic agents interact to force adaptation as conditions change. Darwin and Wallace provided a general mechanism for how this occurs with their theory of natural selection, and we later learned that DNA (deoxyribonucleic acid), a wonderfully simple instructional language written with long chains of pairs of four nucleic acids, was the ultimate scaffold upon which all life forms could be expressed through evolution. The power of that is wonderfully demonstrated by the adaptive radiation of cichlid fish in the rift lakes of Africa (Schedel et al. 2019).
Climatic change is one driver of evolutionary change. We know that climate has changed many times in the past, but generally at a very slow rate relative to our current rate of warming. In fact, all five of earth’s mass extinctions, each eliminating 75-95% of life as it existed then, was caused by some type of climate perturbation. Rare catastrophic events have led to widespread and rapid change, e.g., the meteor that led to the global extinction of most dinosaurs and ammonites along with many other organisms, but even that event was in part caused by secondary climatic effects. Current global climate change deniers often bring up the fact that climate has fluctuated throughout the planet’s existence as evidence for their position. However, humans are now severely impacting the environment both directly and indirectly, and at all conceivable scales. The rate of extinctions over the last century vastly exceeds what would be expected in the absence of human influence, supporting the notion that we have entered a sixth mass extinction. Thus, the Anthropocene, the era dominated by humans, is characterized by unprecedented depletion rates of natural resources and an accelerating increase in CO2 levels. Carbon release is further accelerating as permafrost thaws, releasing vast volumes of trapped CO2. This animation from NOAA shows the variation by latitude and the global rate of increase. The result when combined with all other anthropogenic impacts (habitat destruction, pesticides etc.) is unprecedented loss of biodiversity, prompting media to declare insectageddon and bird apocalypse in response to some recent scientific studies sounding the alarm. (I strongly recommend that you read this and some follow-up posts by Manu Saunders on her excellent blog site “Ecology is not a dirty word” and this by Brian McGill before accepting the media reports at face value.)
When we think of evolution, we generally envision a slow process over many decades, centuries, millennia or even longer. We are of course familiar with resistance evolving to pesticides in insects, and bacteria evolving resistance to antibiotics. Evidence is starting to surface that at least some higher animals are able to evolve fairly rapidly if the selection pressure is strong enough. Humans have had a hand in this for a long time, but for the most part by intentional selection as part of the domestication process of animals and plants. The dog evolving from the wolf into innumerable races that seem to have few, if any, wolf-like characteristics, and crop improvement for increased yield are some familiar examples. Darwin bred fancy pigeons in his attempts to understand the process of natural selection. We have already caused the extinction of many animals through over-exploitation, and this is still a major threat to the continued existence of numerous at-risk species. In addition, humans are now directly causing unintentional very rapid evolutionary adaptation in some animal populations through activities causing extreme selection pressure. This article summarizes the issues quite well, and provides some interesting examples. Below I will highlight a few additional examples that I am aware of.
Tuskless elephants
African elephants have been subject to extensive poaching for their tusks, which are sought after in China for carving and jewellery. Males are particularly sought after because they have larger tusks than females. Females are also killed, however. In some areas, e.g., Mozambiques Gorongosa National Park and Tanzania’s Ruaha National Park, the pressure by poaching on the population has been severe, leading to a much higher level of female tusklessness than in areas with little or no poaching.
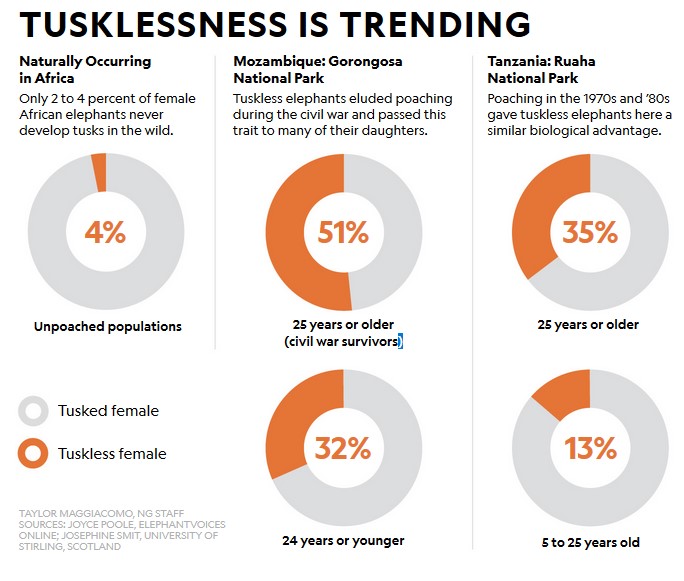
Tusks are used for a number of tasks, but it appears that tuskless females manage well without them. Interestingly, female Indian elephants do not have tusks, possibly as a consequence of historic hunting pressure.
Rapid evolution in sea stars

In 2013 large numbers of sea stars along the Pacific coast of North America were found to essentially disintegrate and die for no apparent reason. The problem persisted into 2014, leading to a rapid decline in the number of sea stars. A paper by Hewson et al. indicated a densovirus as a causal agent, but that is was later confirmed only for the sunflower star, Pycnopodia helianthoides (Hewson et al. 2018). A marine heating event is also implicated, but the ultimate cause of what is now called Sea Star Wasting Syndrome is elusive. Recovery among some populations, e.g., the common ochre sea star, was shown to be due to rapid genetic changes (Schiebelhut et al. 2018). This extremely rapid evolution may indicate that some organisms have the capacity to adapt to environmental changes relatively rapidly, which may be cause for some level of hope.
Bird evolution resulting from anthropogenic activities
Using a data set of over 70,000 migratory birds collected over four decades, most killed by window strikes, Weeks et al. (2020) found that increasing temperatures have led to a decrease in body size, but an increase in wing length. We hypothesize that the increase in wing length is a compensatory adaptation to the reduced body size, which would impact migration due to an increased metabolic cost of flight.
In another study on cliff swallows occupying roadside nesting sites (bridges, overpasses etc.) over three decades, Brown and Bomberger Brown (2013) found that road kill of swallows declined rapidly. Birds that were killed had longer wings than the general population, which the authors hypothesize may be due to selection for short-winged birds that are better able to avoid collisions. They acknowledge that other selection pressures, e.g., severe weather events and a change in the insect fauna, may also have contributed. Nevertheless, their study indicates that evolutionary responses to changing conditions can sometimes occur rapidly.
A particularly interesting example of rapid evolution is the “re-evolution” of an extinct species of rail on Aldabra, a large coral atoll in the Seychelles. A subspecies of the white-throated rail, the Aldabra rail has gone extinct several times due to sea level rise which has submerged the atoll, but has re-emerged subsequent to re-occupation by white-throated rails presumably from Madagascar. White-throated rails are “persistent colonizers”, meaning that they tend to emigrate en masse due to unknown triggers. The Aldabra rail has “re-evolved” in the sense that flight has been lost and the birds have become heavier. The evidence for this comes from comparing fossil and modern humerus bones showing that pre-flood rails were similar to the Aldabra rail as it exists today, whereas post-flood rails were in the process of losing flight. The process took 20,000 years, but that is still rapid in evolutionary terms.
There are many additional examples, but these may be some of the easiest to relate to as the animals are charismatic. Bull and Maron (2016) provide an overview that may be of interest, or you can read this overview for the Reader’s Digest version.
References
Brown, C R and M. Bomberger Brown. 2013. Where has all the road kill gone? Current Biology. 23, R233–R234.
Bull JW and M Maron. 2016. How humans drive speciation as well as extinction. Proceedings of the Royal Society B. 283, 20160600. (doi:10.1098/rspb.2016.0600)
Hewson, I, JB Button, BM Gudenkauf, et al. 2014. Densovirus associated with sea-star wasting disease and mass mortality. PNAS 201416625; DOI: 10.1073/pnas.1416625111
Hewson, I, K Bistolas, SI Kalia et al. 2018. Investigating the Complex Association Between Viral Ecology, Environment, and Northeast Pacific Sea Star Wasting. Frontiers in Marine Science 5: 77 DOI=10.3389/fmars.2018.00077
Hume, JP and D Martill. 2019. Repeated evolution of flightlessness in Dryolimnas rails (Aves: Rallidae) after extinction and recolonization on Aldabra. Zoological Journal of the Linnean Society, 186: 666–672, https://doi.org/10.1093/zoolinnean/zlz018
Schedel, FDB., Z Musilova and UK Schliewen. 2019. East African cichlid lineages (Teleostei: Cichlidae) might be older than their ancient host lakes: new divergence estimates for the east African cichlid radiation. BMC Evolutionary Biology 19: 94. https://doi.org/10.1186/s12862-019-1417-0
Schiebelhut, LM, JB Puritz, and MN Dawson. 2018. Decimation by sea star wasting disease and rapid genetic change in a keystone species, Pisaster ochraceus. Proceedings of the National Academy of Sciences 115(27): 7069-7074; DOI: 10.1073/pnas.1800285115.
Weeks, BC, DE Willard, M Zimova, AA Ellis, ML Witynski, M Hennen, and BM Winger. 2020. Shared Morphological Consequences of Global Warming in North American Migratory Birds. Ecology Letters, 23(2): 316-325. doi: 10.1111/ele.13434.